Leishmaniasis
This article is about human leishmaniasis. For the disease in canids, see canine leishmaniasis.
| Leishmaniasis | |
|---|---|
| Classification and external resources | |
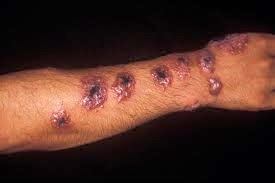
Cutaneous leishmaniasis in the hand of a Central American adult
|
|
| ICD-10 | B55 |
| ICD-9 | 085 |
| DiseasesDB | 3266 29171 3266 7070 |
| MedlinePlus | 001386 |
| eMedicine | emerg/296 |
| MeSH | D007896 |
Infections in humans are caused by more than 20 species of Leishmania.[2] Risk factors include: poverty, malnutrition, deforestation and urbanization.[2] All three types can be diagnosed by directly seeing the parasites under the microscope.[2] Additionally visceral disease can be diagnosed via blood tests.[3]
Leishmaniasis can be partly prevented by sleeping under nets treated with insecticide.[2] Other measures include: spraying insecticides to kill sandflies and treating people with the disease early to prevent further spread.[2] The treatment needed is determined by where the disease is acquired, the species of Leishmania and the type of infection.[2] Some possible medications used for visceral disease include: liposomal amphotericin B,[4] a combination of pentavalent antimonials and paromomycin,[4] and miltefosine.[5] For cutaneous disease paromomycin, fluconazole or pentamidine may be effective.[6]
About 12 million people are currently infected[7] in some 98 countries.[3] There are about 2 million new cases[3] and between 20 to 50 thousand deaths a year.[2][8] About 200 million people in Asia, Africa, South and Central America and southern Europe live in areas where the disease is common.[3][9] The World Health Organization has obtained discounts on some medications to treat the disease.[3] The disease may occur in a number of other animals including dogs and rodents.[2]
Signs and symptoms
The symptoms of leishmaniasis are skin sores which erupt weeks to months after the person is bitten by infected sand flies.Leishmaniasis may be divided into the following types:[10]
- Cutaneous leishmaniasis is the most common form, which causes an open sore at the bite sites, which heals in a few months to a year and half, leaving an unpleasant-looking scar.[2][3] Diffuse cutaneous leishmaniasis produces widespread skin lesions which resemble leprosy, and may not heal on its own.[3]
- Mucocutaneous leishmaniasis causes both skin and mucosal ulcers with damage primarily of the nose and mouth.[2][3]
- Visceral leishmaniasis or kal-azar is the most serious form, and is potentially fatal if untreated.[2] Other consequences, which can occur anywhere from a few months to years after infection, include fever, damage to the spleen and liver, and anemia.[2]
Cause
Leishmaniasis is transmitted by the bite of infected female phlebotomine sandflies[2] which can transmit the infection Leishmania.[2] The sandflies inject the infective stage, metacyclic promastigotes, during blood meals (1). Metacyclic promastigotes that reach the puncture wound are phagocytized by macrophages (2) and transform into amastigotes (3). Amastigotes multiply in infected cells and affect different tissues, depending in part on which Leishmania species is involved (4). These differing tissue specificities cause the differing clinical manifestations of the various forms of leishmaniasis. Sandflies become infected during blood meals on infected hosts when they ingest macrophages infected with amastigotes (5,6). In the sandfly's midgut, the parasites differentiate into promastigotes (7), which multiply, differentiate into metacyclic promastigotes, and migrate to the proboscis (8).The genomes of three Leishmania species (L. major, L. infantum, and L. braziliensis) have been sequenced and this has provided much information about the biology of the parasite. For example, in Leishmania, protein-coding genes are understood to be organized as large polycistronic units in a head-to-head or tail-to-tail manner; RNA polymerase II transcribes long polycistronic messages in the absence of defined RNA pol II promoters, and Leishmania has unique features with respect to the regulation of gene expression in response to changes in the environment. The new knowledge from these studies may help identify new targets for urgently needed drugs and aid the development of vaccines.[11]
Vector
Although most of the literature mentions only one genus transmitting Leishmania to humans (Lutzomyia) in the US, a 2003 study by Galati suggested a new classification for American sand flies, elevating several subgenera to the genus level. Elsewhere in the world, the genus Phlebotomus is considered the vector of leishmaniasis.[11]Organisms
Visceral disease is usually cause by Leishmania donovani, Leishmania infantum or Leishmania chagasi.[3] But occasionally these species may cause other forms of disease.[3] The cutaneous form of the disease is caused by more than 15 types of Leishmania.[3]Risk factors
Risk factors include: poverty, malnutrition, deforestation and urbanization.[2]Diagnosis
Leishmaniasis is diagnosed in the hematology laboratory by direct visualization of the amastigotes (Leishman-Donovan bodies). Buffy-coat preparations of peripheral blood or aspirates from marrow, spleen, lymph nodes, or skin lesions should be spread on a slide to make a thin smear and stained with Leishman's or Giemsa's stain (pH 7.2) for 20 minutes. Amastigotes are seen with monocytes or, less commonly in neutrophils, of peripheral blood and in macrophages in aspirates. They are small, round bodies 2–4 μm in diameter with indistinct cytoplasm, a nucleus, and a small, rod-shaped kinetoplast. Occasionally, amastigotes may be seen lying free between cells.[12] However, the retrieval of tissue samples is often painful for the patient and it can be difficult to identify the infected cells. For these reasons, other indirect immunological methods of diagnosis are developed. These methods include the enzyme-linked immunosorbent assay (ELISA), antigen coated dipsticks, and the direct agglutination test (DAT). Although these tests are readily available, they are not the standard diagnostic tests due to their insufficient sensitivity and specificity.There are several different polymerase chain reaction (PCR) tests for the detection of Leishmania DNA.[3] With the PCR assay, a specific and sensitive diagnostic procedure is finally possible.
Most forms of the disease are transmitted only from non-human animals, but some can be spread between humans. Infections in humans are caused by about 21 of 30 species that infect mammals[citation needed]: the different species look the same, but they can be differentiated by isoenzyme analysis, DNA sequence analysis, or monoclonal antibodies.
Prevention
Leishmaniasis can be partly prevented by using nets treated with insecticide while sleeping.[2]Treatment
The evidence around the treatment of cutaneous leishmaniasis is poor.[3] A number of topical treatments may be used for cutaneous leishmaniasis. Which treatments are effective depends on the strain, with topical paromomycin effective for L. major, L. tropica, L. mexicana, L. panamensis and L. braziliensis.[6] Pentamidine is effective for L. guyanensis.[6] Oral fluconazole or itraconazole appears effective in L. major and L. tropica.[3][6]
Epidemiology
Cutaneous leishmaniasis in North Africa; Leishmania infantum = green, Leishmania major = blue, Leishmania tropica = red[13]
Disability-adjusted life year for leishmaniasis per 100,000 inhabitants.
no data
less than 20
20–30
30–40
40–50
50–60
60–70
70–80
80–100
100–120
120–150
150–200
more than 200
Leishmaniasis is found through much of the Americas from northern Argentina to southern Texas, though not in Uruguay or Chile, and has recently been shown to be spreading to North Texas.[17] Leishmaniasis is also known as papalomoyo, papa lo moyo, ulcero de los chicleros and "chiclera" in Latin America.[18] During 2004, an estimated 3,400 troops from the Colombian army, operating in the jungles near the south of the country (in particular around the Meta and Guaviare departments), were infected with leishmaniasis. Apparently, a contributing factor was that many of the affected soldiers did not use the officially provided insect repellent, because of its allegedly disturbing odor. Nearly 13,000 cases of the disease were recorded in all of Colombia throughout 2004, and about 360 new instances of the disease among soldiers had been reported in February 2005.[19][20][21]
The disease is found across much of Asia, and in the Middle East. Within Afghanistan, leishmaniasis occurs commonly in Kabul, partly due to bad sanitation and waste left uncollected in streets, allowing parasite-spreading sand flies an environment they find favorable.[22][23] In Kabul, the number of people infected was estimated to be at least 200,000, and in three other towns (Herat, Kandahar and Mazar-i-Sharif) there were about 70,000 more, according to WHO figures from 2002.[24][verification needed] Kabul is estimated as the largest center of cutaneous leishmaniasis in the world, with approximately 67,500 cases as of 2004.[25]
Africa, in particular the East and North,[13] is home to cases of leishmaniasis. The disease is spreading to Southern Europe, but is not found in Australia or Oceania.[citation needed]
Leishmaniasis is mostly a disease of the developing world, and is rarely known in the developed world outside a small number of cases, mostly in instances where troops are stationed away from their home countries. Leishmaniasis has been reported by U.S. troops stationed in Saudi Arabia and Iraq since the Gulf War of 1990, including visceral leishmaniasis.[26][27][28] In September 2005, the disease was contracted by at least four Dutch marines who were stationed in Mazari Sharif, Afghanistan, and subsequently repatriated for treatment.[citation needed]
History
A 1917 case of cutaneous leishmaniasis in the Middle East, known then
locally as "Jericho Buttons" for the frequency of cases near the ancient
city of Jericho
It remains unclear who first discovered the organism. Surgeon Major Cunningham of the British Indian army possibly saw it first in 1885 without being able to relate it to the disease.[31][32] Peter Borovsky, a Russian military surgeon working in Tashkent, conducted research into the etiology of oriental sore, locally known as "Sart sore", and in 1898 published the first accurate description of the causative agent, correctly described the parasite's relation to host tissues and correctly referred it to Protozoa. However, because his results were published in Russian in a journal with low circulation, his priority was not internationally acknowledged during his lifetime.[33] In 1901, Leishman identified certain organisms in smears taken from the spleen of a patient who had died from "dum-dum fever" (Dum Dum is an area close to Calcutta) and proposed them to be trypanosomes, found for the first time in India.[34] A few months later, Captain Charles Donovan (1863–1951) confirmed the finding of what became known as Leishman-Donovan bodies in smears taken from patients in Madras in southern India.[35] But it was Ronald Ross who proposed that Leishman-Donovan bodies were the intracellular stages of a new parasite, which he named Leishmania donovani.[36] The link with the disease kala-azar was first suggested by Charles Donovan, but was conclusively demonstrated by Charles Bentley's discovery of Leishmania donovani in patients with kala-azar.[37] The disease became a major problem for Allied troops fighting in Sicily during the Second World War; research by Leonard Goodwin then showed pentostam was an effective treatment.[38]
Society and culture
The Institute for OneWorld Health has reintroduced the drug paromomycin for treatment of leishmaniasis, results with which led to its approval as an orphan drug. The Drugs for Neglected Diseases Initiative is also actively facilitating the search for novel therapeutics. A treatment with paromomycin will cost about $10. The drug had originally been identified in 1960s, but had been abandoned because it would not be profitable, as the disease mostly affects poor people.[39] The Indian government approved paromomycin for sale in August 2006.[40]The World Health Organization has gotten a reduced cost for liposomal amphotericin B at $18 a vial, however many vials may be needed for treatment and it must be kept cool.[3]
Research
On February 2012, the nonprofit Infectious Disease Research Institute launched the world’s first human clinical trial of the visceral leishmaniasis vaccine. The vaccine is a recombinant form of two fused Leishmania parasite proteins with an adjuvant. Two phase 1 clinical trials with healthy volunteers are to be conducted. The first one takes place in Washington (state) and is followed by a trial in India.[43]
In 2009, the Hebrew University of Jerusalem Kuvin Center for the Study of Infectious and Tropical Diseases, in a collaborative effort with Addis Ababa University, was awarded a grant by the Bill & Melinda Gates Foundation for research into visceral leishmaniasis in Ethiopia. The project will gather data to be analyzed to identify the weak links in the transmission cycle, and devise methods for control of the disease.[44]
HIV protease inhibitors have been found to be active against Leishmania species in two in vitro studies in Canada and India. The studies reported the intracellular growth of parasites was controlled by nelfinavir and ritonavir in a human monocyte cell line and also in human primary monocyte-derived macrophages.[45]
Since September 2011 there exists a World Community Grid project called Drug Search for Leishmaniasis which has the goal to find new drugs against this disease.[46]
Notable cases
Marguerite Higgins, Pulitzer Prize winning journalist, died in early 1966 from leishmaniasis contracted while on an assignment the previous year.Magazine photographer Joel Sartore was diagnosed with the disease after a skin lesion refused to heal following a photo shoot in the Bolivian wilderness. Following intensive IV treatment similar to chemotherapy, his infection has resolved.[47]
While filming the latest series of Extreme Dreams in Peru, UK television presenter Ben Fogle caught the disease. He was left bedridden for three weeks on his return home. Fogle was treated at London's Hospital for Tropical Diseases.[48]
During his two-and-a-half-year walk through the Amazon, Ed Stafford tested positive for cutaneous leishmaniasis in Oriximiná, Para, Brazil. He convinced the local doctor that he could not stay in one place for treatment due to his undertaking and was prescribed with 20 days of intravenous injections that he self-administered in the jungle.












No comments:
Post a Comment